|
EndoBarrier Worldwide Registry
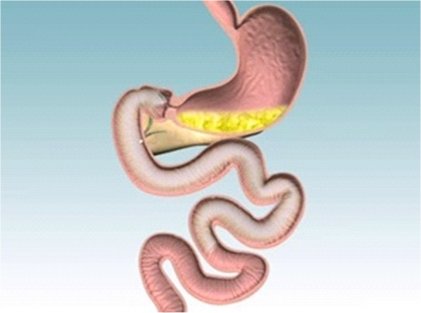
EndoBarrier is a 60cm tube-like liner
made from a thin, flexible and durable impermeable polymer. It is
inserted by an endoscopy and prevents food that passes through it from
contacting the first two feet of small bowel intestine. In clinical
trials it leads to weight loss and improved glycaemic control and may
become a useful treatment option in the future.
ABCD and EndoBarrier
ABCD is currently funding the
REVISE-Diabesity
and
End-OSA EndoBarrier studies, which are supported by the NHS, as National Institute for
Health Research’ (NIHR) Clinical (Diabetes) Research Network Portfolio
Studies. One year data from the REVISE study was presented at the
ADA 2016
as well as at the
EASD in 2016, and there has been a
BBC
news item sparked by this data. There was also a
BBC
news item following the launch of the End-OSA study of EndoBarrier
in diabetes with obstructive sleep apnoea.
Need for a registry
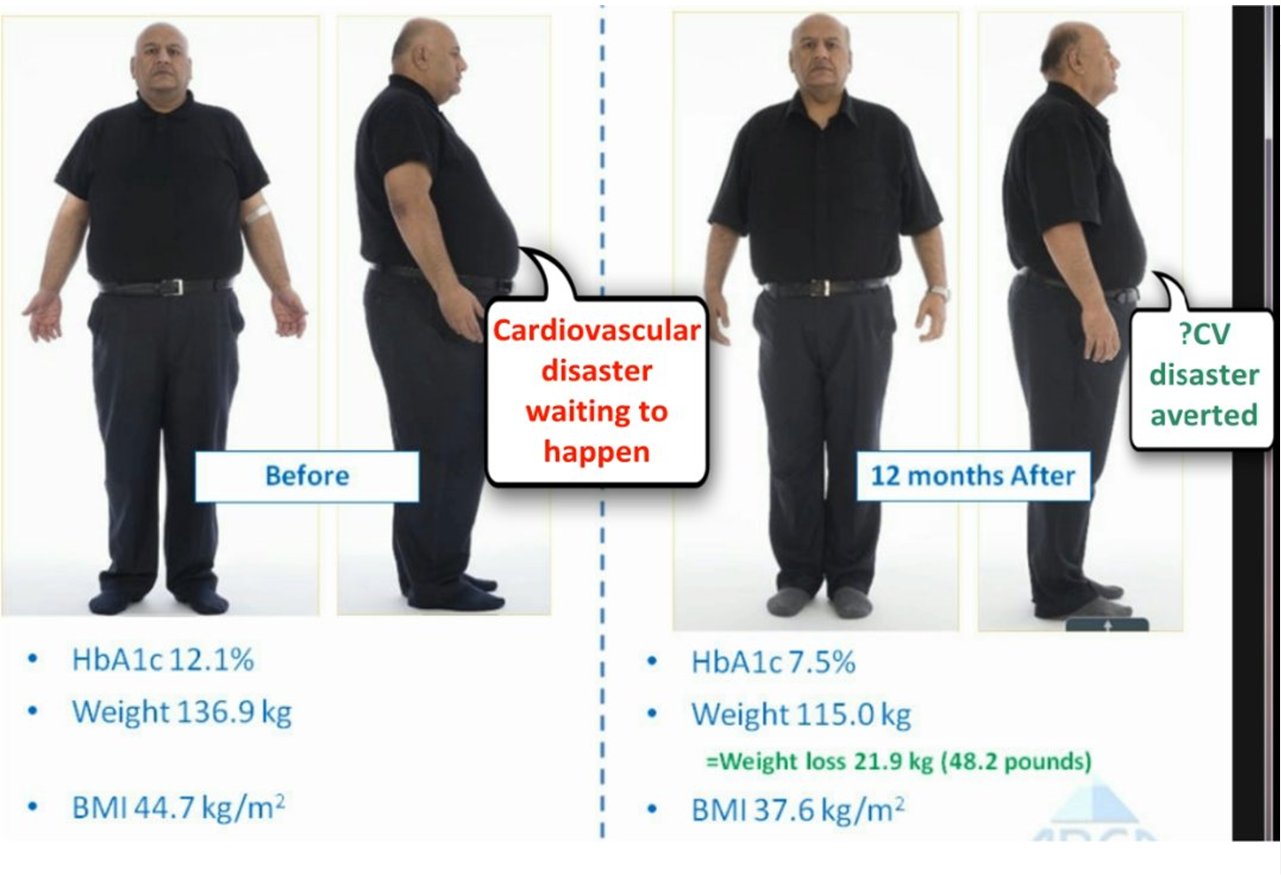
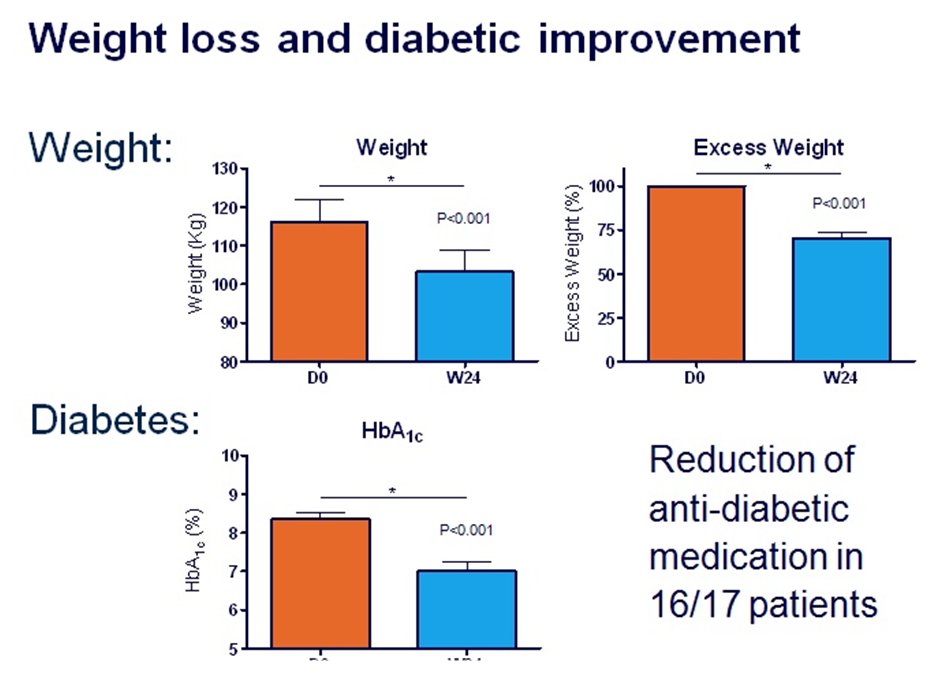
Over 3000 patients in the world have had EndoBarrier treatment. Those of
us who have used it have found that most patients gain considerable
improvement in terms of reduction in weight, improvement in diabetes
control, reduction in the need for other diabetes medications, in
particular insulin, improvement in patient wellbeing and examples from
the experience in Birmingham, UK, are shown
here.
Side effects, such as they are, seem to be acceptable considering the
benefit. If the information about all these patients could be collected
into a single worldwide registry we would have enormous power to
demonstrate the benefits of EndoBarrier in relation to the risks. Using
the
technology already established for the ABCD nationwide and worldwide
audits, ABCD has created an independent
online tool
to collect this data.
Local analysis
The tool facilitates Centres entering data to analyse their own data
very easily for the purposes of presentation and publication, etc.
National registries
The
German national registry data was presented at the EASD in 2016. We
introduce here
the independent worldwide EndoBarrier registry tool which now allows
other nations to easily follow suit and analyse their own national data
for the purposes of presentation and publication, etc: The tool very
easily allows all the Centres in a Country to combine their data.
Worldwide registry
The tool easily then allows worldwide analysis of all the data in
the world and it allows for comparison between countries
Paper forms
If you would like to collect data on paper forms that exactly match the
on-line tool then you can download for printing the
first visit form and the
follow up visit form
Ownership of the data
As well established for the other ABCD national and worldwide audits of
new diabetes therapies, the data is jointly owned by all those
contributing to the registry and all contributors will be mentioned in
all publications utilising the worldwide data. On the safety side the
tool will allow us to know the exact frequency of adverse events and the
circumstances in which they occur
Registration
All EndoBarrier using Centres are therefore encourage to register and
enter the data from their EndoBarrier treated patients. Data can be
added retrospectively or prospectively.
Click here to register
and join the
registry |